 Dr. Greger said this in his book, How Not To Die:
Dr. Greger said this in his book, How Not To Die:
Thousands of published studies seem to have demonstrated that antidepressant drugs are effective. The key word here, though, may be published. What if drug companies decided to publish only those studies that showed a positive effect but quietly shelved and concealed any studies showing the drugs didn’t work? To find out if this was the case, researchers applied to the Food and Drug Administration under the U.S. Freedom of Information Act (FOIA) to get access to the published and unpublished studies submitted by pharmaceutical companies. What they found was shocking.
According to the published literature, the results of nearly all antidepressant trials were positive. In contrast, FDA analysis of trial data – including the unpublished studies – demonstrated that roughly half of the trials showed the drugs didn’t work after all. When all the data- published and unpublished – were combined, antidepressants failed to show a clinically significant advantage over placebo sugar pills. This finding suggests that the placebo effect explains the apparent clinical effectiveness of antidepressants. In other words, improvements in mood may be a result of the patient’s belief in the power of the drug – not the drug itself.
That can’t be true, can it? Yes, it can. Here’s the study he referenced:
Selective Publication Of Antidepressant Trials And Its Influence On Apparent Efficacy, New England Journal of Medicine, January 2008
And here’s its incriminating chart:
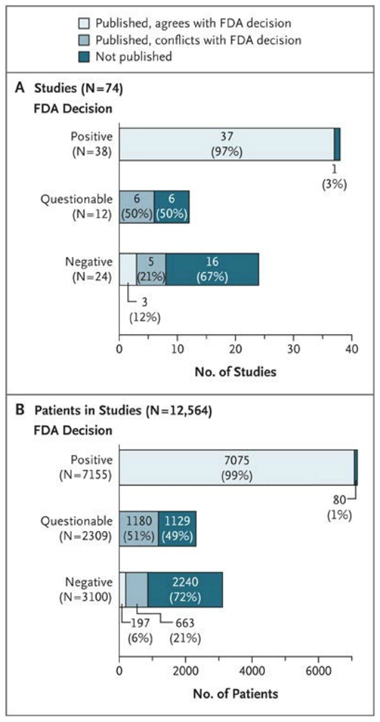
Figure 1. Effect of FDA Regulatory Decisions on Publication.
Among the 74 studies reviewed by the FDA (Panel A), 38 were deemed to have positive results, 37 of which were published with positive results; the remaining study was not published. Among the studies deemed to have questionable or negative results by the FDA, there was a tendency toward nonpublication or publication with positive results, conflicting with the conclusion of the FDA.Among the 12,564 patients in all 74 studies (Panel B), data for patients who participated in studies deemed positive by the FDA were very likely to be published in a way that agreed with the FDA. In contrast, data for patients participating in studies deemed questionable or negative by the FDA tended either not to be published or to be published in a way that conflicted with the FDA’s judgement.
There’s another chart that is too big for me to post here (but you can click through to see it) that breaks publication down by individual study and by drug. Same thing. Studies that showed the drug didn’t work didn’t get published. And there were a lot of them. The authors concluded:
Selective reporting of clinical trial results may have adverse consequences for researchers, study participants, health care professionals, and patients.
These aren’t obscure drugs. They include: Paxil, Prozac, Zoloft, Wellbutrin, Celexa, Cymbalta, Effexor, and others.
How many other drugs are showing ineffectiveness, or worse, harm, in research that never sees the light of day? Has publication bias allowed published research to become just another form of advertising?

I may have said this before, but there was a hilarious routine once on NPR about the benefits of taking Placebo for what ails you! http://www.npr.org/2010/12/23/132291795/This-Placebo-Could-Be-The-Drug-For-You
LikeLike
That’s funny.
It’s amazing to me how powerful a placebo effect can be. Even when we know!
LikeLike